Part 1 and part 2 introduced mass spectrometry-based proteomic methods, like PTMScan® technology, for the study of post-translational modifications (PTMs). In the final part of this series you can see how PTMScan can be applied in translational research.
Personalized Medicine and the Power of Proteomics
We’re living at a time when personalized medicine is possible. That is, treatment of diseases like cancer can be tailored to each individual rather than treating with a general treatment strategy that might or might not have worked in the past for other patients. Personalized medicine increases the chance of a positive outcome while hopefully decreasing side effects, which can make a big difference to the quality of the patient’s life. This targeted approach is largely made possible because of recent advances in genomic and proteomic technologies. Proteomic methods like PTMScan can provide insight into disease biology that is not feasible at the genetic level. For example, you can analyze protein activation or inhibition caused by PTMs such as phosphorylation, methylation, and acetylation.
Now let’s look at a bench-to-bedside story for a specific example of how powerful these methods can be...
 Case Study: Identifying Disease Drivers in NSCLC
Case Study: Identifying Disease Drivers in NSCLC
Some cancers rely on the activity of tyrosine kinases like EGFR, c-Met, and others for their growth and survival. These proteins are called disease drivers. To identify novel disease drivers, CST performed a global survey of tyrosine kinase activity in non-small cell lung cancer (NSCLC) (1). This was done with the hope that if we understand what active proteins these cancer cells depend on, we can inhibit those proteins and stop the growth of the cancer cells.
CST scientists carried out PTMScan by using a phospho-tyrosine PTM-antibody to immunoaffinity enrich phosphorylated peptides from 41 NSCLC cell lines and 150 NSCLC tumors. They then analyzed the enriched peptide population using LC-MS/MS. This analysis allowed CST scientists to identify phosphorylation at 4551 tyrosine residues on more than 2700 proteins, including many tyrosine kinases and tyrosine kinase substrates, dramatically increasing our understanding of the tyrosine phosphoproteome in NSCLC. The identified proteins were ranked according to their degree of phosphorylation. The tyrosine kinases Met, ALK (anaplastic lymphoma kinase), ROS, PDGFRa, DDR1, and EGFR were among the top 10 candidates for follow-up with high phosphorylation levels found in both cell lines and tumors.
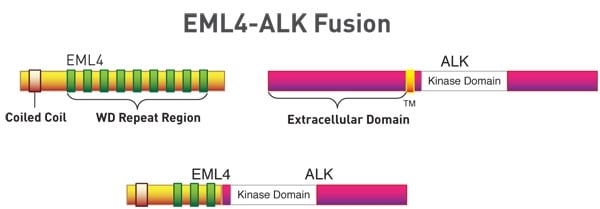
Further investigation of ALK in these cells and tumors revealed fusion of the N-terminus of EML4 (echinoderm microtubule-associated protein-like 4), a microtubule-associated protein, with the C-terminus of ALK, which contains the kinase domain. This fusion protein is highly oncogenic, and it turns out that 3-7% of patients with NSCLC express the fusion protein in their tumors (1-4). Thus, for these patients the EML4-ALK fusion may be driving the oncogenic progress of their disease. Cancer cells expressing the fusion protein are, in fact, sensitive to the small molecule ALK inhibitor crizotinib, and in 2011 the FDA approved crizotinib for the treatment of ALK-positive NSCLC.
Following this study, CST developed a highly specific and sensitive antibody, ALK (D5F3®) XP® Rabbit mAb #3633, which detects full-length ALK and the ALK-EML4 fusion protein. Currently, the gold standard for detecting ALK rearrangements in patients is fluorescence in situ hybridization, but this is expensive, requires specialized training, is labor-intensive, and is therefore not easily scalable. The FDA recently approved an immunohistochemistry (IHC) companion diagnostic test, which uses the ALK D5F3 clone licensed from CST (6). This will help physicians to easily pinpoint which NSCLC patients may be effectively treated with crizotinib.
A similar approach has been used to investigate tyrosine phosphorylation in other types of cancer such as ovarian cancer where other abnormally active ALK fusion proteins were identified and found to be sensitive to crizotinib (7).
Why PTM Proteomics Matters for Disease Research
As you can see from simplifying proteomics parts 1, 2, and 3, immunoaffinity enrichment followed by mass spectrometry is an ideal way to identify and quantify even the lowest abundance PTM peptides from a complex biological sample. This makes methods like PTMScan extremely powerful tools for quickly identifying biomarkers of disease, identifying and validating drug targets, elucidating off-target effects, exploring drug mechanism of action, and ultimately facilitating our ability to diagnose and treat disease.
Download the Simplifying Proteomics Brochure to learn about:
- The types of antibodies used in PTMScan
- PTMScan Discovery vs PTMScan Direct and how to decide which is right for your research
- PTMScan products and services
Cell Signaling Technology provides products and services are for research use only and
not for use in diagnostic or therapeutic procedures.
Select References
- Rikova K, Guo A, Zeng Q, Possemato A, Yu J, Haack H, Nardone J, Lee K, Reeves C, Li Y, Hu Y, Tan Z, Stokes M, Sullivan L, Mitchell J, Wetzel R, Macneill J, Ren JM, Yuan J, Bakalarski CE, Villen J, Kornhauser JM, Smith B, Li D, Zhou X, Gygi SP, Gu TL, Polakiewicz RD, Rush J, Comb MJ (2007) Global survey of phosphotyrosine signaling identifies oncogenic kinases in lung cancer. Cell 131(6), 1190–203.
- Soda M, Choi YL, Enomoto M, Takada S, Yamashita Y, Ishikawa S, Fujiwara S, Watanabe H, Kurashina K, Hatanaka H, Bando M, Ohno S, Ishikawa Y, Aburatani H, Niki T, Sohara Y, Sugiyama Y, Mano H (2007) Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature 448(7153), 561–6.
- Koivunen JP, Mermel C, Zejnullahu K, Murphy C, Lifshits E, Holmes AJ, Choi HG, Kim J, Chiang D, Thomas R, Lee J, Richards WG, Sugarbaker DJ, Ducko C, Lindeman N, Marcoux JP, Engelman JA, Gray NS, Lee C, Meyerson M, Jänne PA (2008) EML4-ALK fusion gene and efficacy of an ALK kinase inhibitor in lung cancer. Clin. Cancer Res. 14(13), 4275–83.
- Shaw AT, Yeap BY, Mino-Kenudson M, Digumarthy SR, Costa DB, Heist RS, Solomon B, Stubbs H, Admane S, McDermott U, Settleman J, Kobayashi S, Mark EJ, Rodig SJ, Chirieac LR, Kwak EL, Lynch TJ, Iafrate AJ (2009) Clinical features and outcome of patients with non-small-cell lung cancer who harbor EML4-ALK. J. Clin. Oncol. 27(26), 4247–53.
- Press Release: Ventana ALK IHC Test Gets FDA Approval as CDx for Pfizer's Xalkori
- Ren H, Tan ZP, Zhu X, Crosby K, Haack H, Ren JM, Beausoleil S, Moritz A, Innocenti G, Rush J, Zhang Y, Zhou XM, Gu TL, Yang YF, Comb MJ (2012) Identification of anaplastic lymphoma kinase as a potential therapeutic target in ovarian cancer. Cancer Res. 72(13), 3312–23.