During hematopoiesis, myeloid cells develop from a common myeloid progenitor (CMP) found in the bone marrow. This lineage—which includes monocytes, granulocytes, erythrocytes, and platelets—is a primary component of the innate immune system and serves as a first line of defense against infection. The study of myeloid cell development and function is a key focus in immunology research, as these cells play critical roles in immune responses and disease.
Related: Immunology: What are lymphoid cells and how are they identified?
This blog explores the main types of myeloid cells as well as key markers used to identify them.
Types of Myeloid Cells
Researchers have identified a number of distinct subtypes of myeloid cells, many of which can be further subcategorized into different functional states, and each playing a unique role in the immune response. The ability to accurately identify and quantify these subtypes is essential for understanding which cell populations are recruited and activated in response to certain pathogens and how they contribute to the resolution of immune challenges.

A few different methods can be used to distinguish myeloid lineage cell types. At a basic level, myeloid cell subsets can be identified based on their morphology and distribution within tissues or blood. However, full classification typically requires immunophenotyping, a technique that leverages antibodies to detect the expression of distinct cell surface molecules, which can be performed using assays such as flow cytometry or immunohistochemistry (IHC).
Immunophenotyping generally involves using panels of antibodies that recognize specific cluster of differentiation (CD) markers—cell surface molecules expressed by different cell types at various stages of differentiation. By analyzing CD marker expression patterns, researchers can detect and quantify the presence of specific immune cells within a mixed population.
|
|
Explore related CST antibody sampler kits, which contain antibodies to many of the markers mentioned in this blog: • Mouse Reactive M1 vs M2 Macrophage IHC Antibody Sampler Kit #97624 • Human Reactive M1 vs M2 Macrophage IHC Antibody Sampler Kit #84223 • Suppressive Myeloid Cell Phenotyping IHC Antibody Sampler Kit #86791 |
|
Functional Overview of Myeloid Cells
CMPs develop into a stunning array of terminally differentiated myeloid cell types. Major branches within this lineage include:
-
Megakaryocytes, which produce the platelets necessary for normal blood clotting.
-
Erythrocytes, or red blood cells, which are responsible for carrying oxygen to tissues.
-
Granulocytes, which include basophils, eosinophils, mast cells, and neutrophils.
-
Monocytes, which serve as progenitors for macrophages and dendritic cells.
Each type of myeloid cell participates in the immune response in a distinct way. For example, basophils contribute to many inflammatory reactions and release heparin, an anticoagulant that slows blood clot formation. Eosinophils are known for their role in defending against parasitic and viral infections, primarily through the release of major basic proteins and ribonucleases. Neutrophils, the most abundant type of granulocyte, act as first responders at sites of inflammation and infection, where they attack and remove invading microorganisms through phagocytosis, degranulation, and formation of neutrophil extracellular traps, or NETs.
When tissues are injured or infected by a pathogen, monocytes in the blood are recruited to the affected tissue, where they may differentiate into macrophages. After differentiation, macrophages detect foreign antigens and damaged cells and destroy them via phagocytosis. Upon completion of this process, macrophages can also process and present antigens to T cells, leading to their activation.
Dendritic cells also use phagocytosis to engulf cellular and foreign material, which they process and present as antigens to T cells. While other types of immune cells can present antigens to T cells, dendritic cells, along with macrophages and B cells, execute this function far more frequently and are known as professional antigen-presenting cells (APCs). APCs serve as critical links to relay information about pathogens between the innate immune system and adaptive immune system.
Myeloid Lineage Cell Markers
Accurately identifying myeloid cell subsets relies on the use of well-characterized phenotyping markers. While the expression of some markers is restricted to specific immune cell lineages, allowing for phenotyping with a single readout, analysis of multiple markers is generally required to determine the phenotype and maturation state of myeloid cells due to overlapping expression patterns.
For example, CD11b can be used as a broad, pan-myeloid marker, but additional readouts are needed to identify subsets and functional states.
- Macrophage Markers: The most common marker for mouse macrophages is F4/80. Human macrophages express CD68 and HLA-DR. Upon differentiation into M1-like or M2-like macrophages, however, these cells can express additional markers, such as CD80 or CD206, respectively.
 IHC analysis of paraffin-embedded mouse liver using F4/80 (D2S9R) Rabbit Monoclonal Antibody #70076.
IHC analysis of paraffin-embedded mouse liver using F4/80 (D2S9R) Rabbit Monoclonal Antibody #70076.
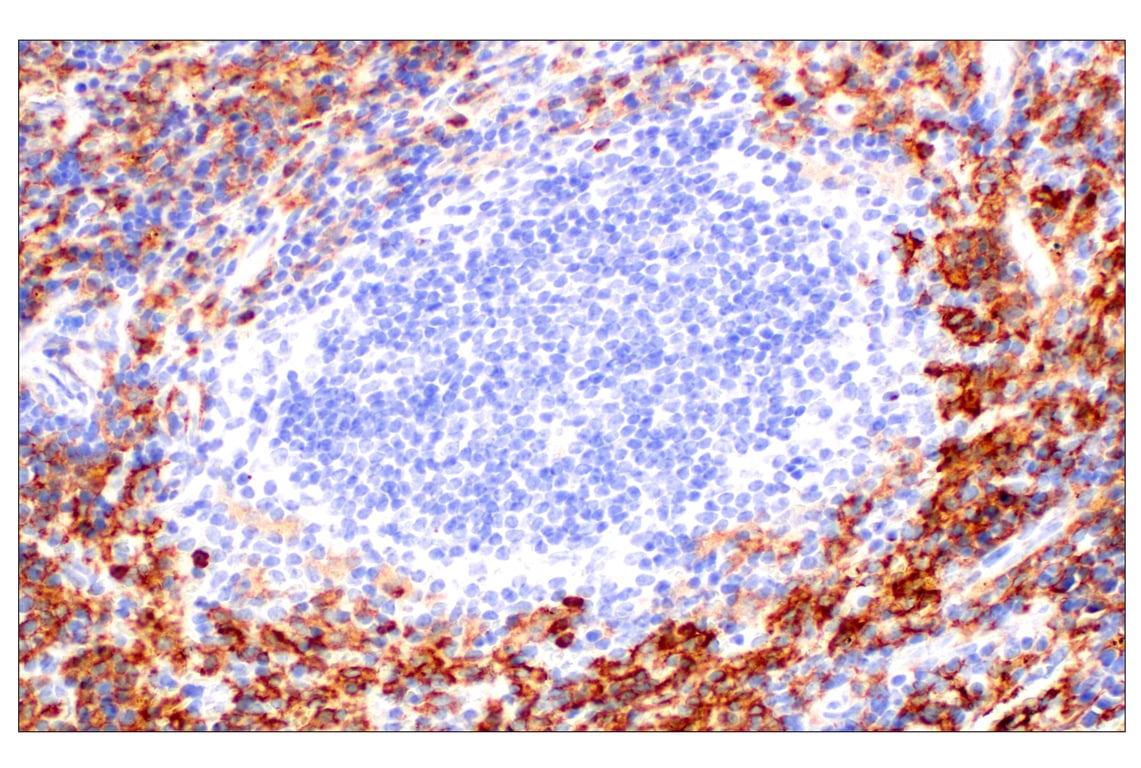
- Dendritic Cell Markers: CD11c is a common marker of conventional dendritic cells (cDCs). Similar to macrophages, however, dendritic cells express different markers after differentiating into cDC1 or cDC2. For example, cDC1s can be identified by their expression of XCR1, while cDC2s can be distinguished by their expression of SIRPα.
 IHC analysis of paraffin-embedded mouse spleen using recombinant monoclonal antibody CD11c (D1V9Y) Rabbit mAb #97585.
IHC analysis of paraffin-embedded mouse spleen using recombinant monoclonal antibody CD11c (D1V9Y) Rabbit mAb #97585.
- Neutrophil Markers: Human neutrophils can be distinguished from other myeloid cells based on their expression of CD16 and CD66b or CD15/SSEA1. Mature neutrophils also express CD10. In mice, Ly-6G is commonly used as a pan-neutrophil marker.
 IHC analysis of paraffin-embedded normal rhesus monkey spleen using CD16 (D1N9L) Rabbit Monoclonal Antibody #24326.
IHC analysis of paraffin-embedded normal rhesus monkey spleen using CD16 (D1N9L) Rabbit Monoclonal Antibody #24326.
- Mast Cell Markers: While both mast cells and basophils express FcεRIA, the combined expression of FcεRIA and CD117 (also known as c-kit) is unique to mast cells. In humans, mast cells can also be identified by their high expression of tryptase, a granule protein. When activated, human mast cells also express CD203c (also known as ENPP3).
 IHC analysis of paraffin-embedded human squamous cell lung carcinoma using c-Kit (D3W6Y) Rabbit Monoclonal Antibody #37805.
IHC analysis of paraffin-embedded human squamous cell lung carcinoma using c-Kit (D3W6Y) Rabbit Monoclonal Antibody #37805.
- Basophil Markers: Basophils can be identified by their expression of FcεRIA and lack of CD117 (c-kit).
- Eosinophil Markers: The combination of CD193 and Siglec-8 expression, and absence of CD16 expression, can be used to identify human eosinophils. Mouse eosinophils express the combination of CD193, F4/80, and Siglec-F.
Immune Cell Marker Pathway Guides
Need a quick reference guide for immune cell markers? Download our comprehensive immune cell marker pathway guides to explore human and mouse markers, as well as relevant CST antibody products validated for IHC and flow cytometry.
Updated May 2025. Originally published May 2020. CST Content Marketing Manager Alexandra Foley, and Tamar Tamar Aprahamian, PhD, founder of JetPub Scientific Communications, contributed to this blog.25-ICT-56550









