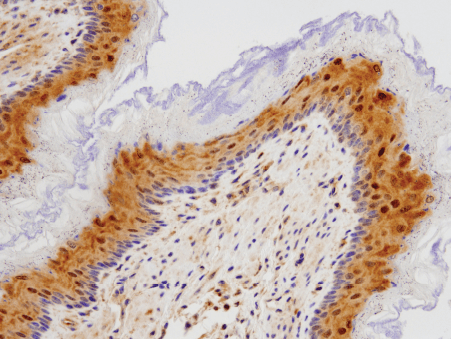
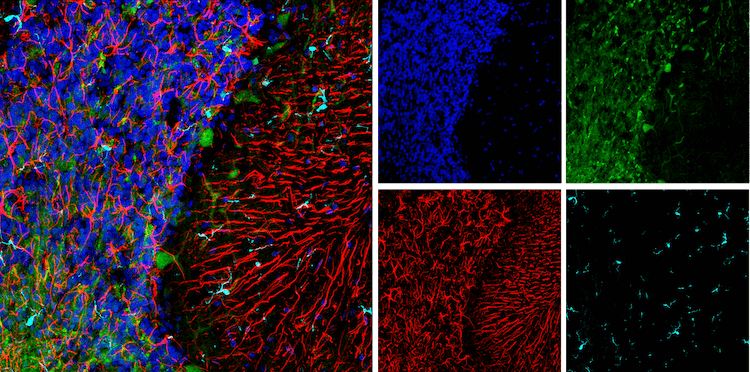
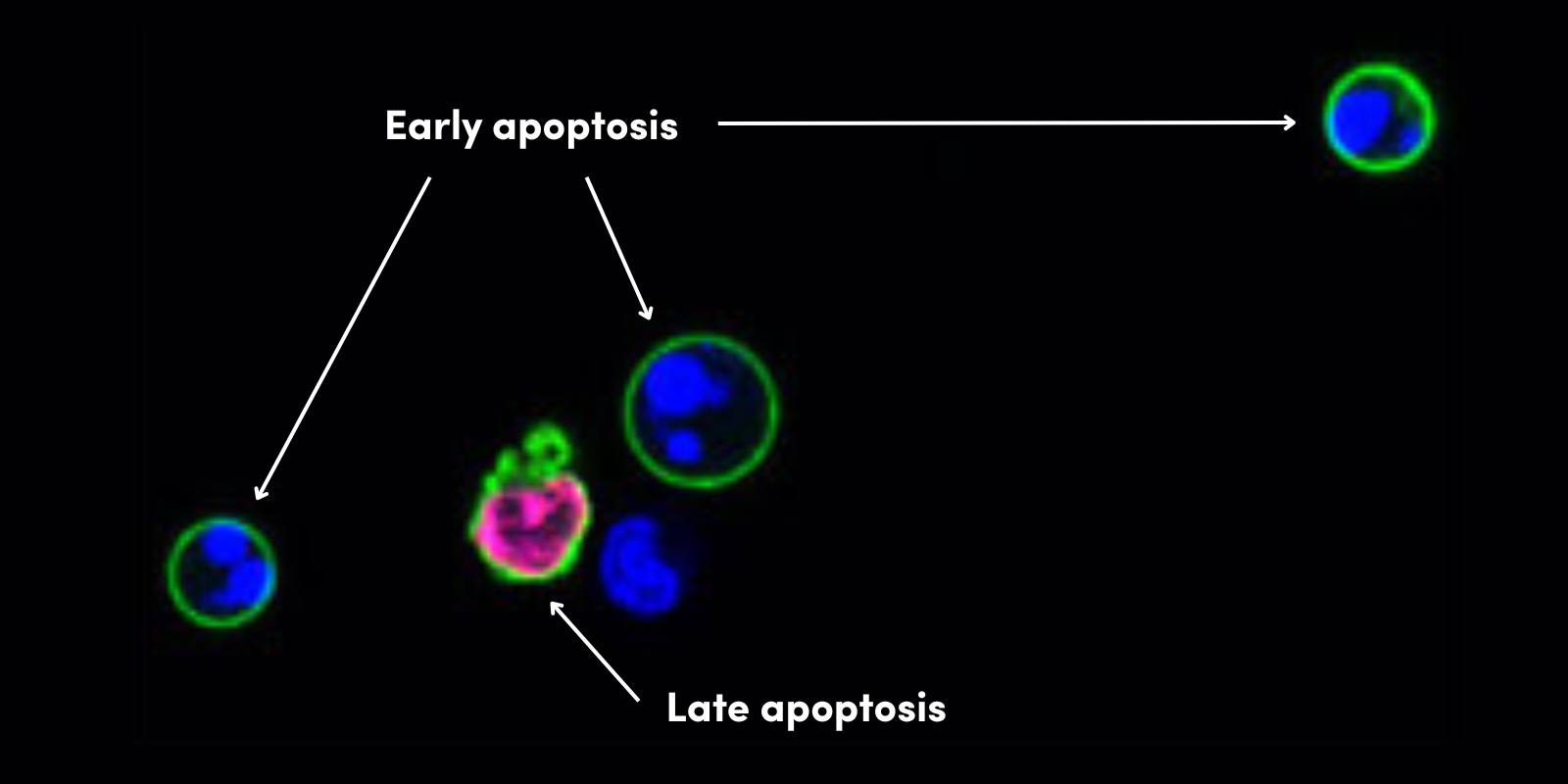
During this time of crisis, healthcare providers and researchers around the world are racing to develop SARS-CoV-2 testing methods. Testing strategies include PCR to diagnose active infection (by testing for the presence of viral genetic material) and the use of antibody test kits on blood (also known as serological tests), in order to track exposure and possible immunity in the population (1,2,3). With these diagnostic tools, the scientific community aims to get a better understanding of the scale and trajectory of the SARS-CoV-2 pandemic.

However, scientists from international research communities, including the UK and Spain, have indicated that many serological antibody test kits provided for evaluation have “not performed well” according to John Bell, Professor of Medicine at Oxford (1). Bell further noted that there were too many false negatives and/or false positives, leading to inaccurate representation of epidemiological data including viral exposure and possible immunity (1). In the U.S. concern for the reliability and availability of the specific antibody serological tests was echoed by members of the National Academy of Sciences’ Standing Committee on Emerging Infectious Diseases and 21st Century Health threats (4). In a letter to the White House, the NAS scientists noted that results from the current antibody tests “should be viewed as suspect until rigorous controls are performed and performance characteristics described” (4).
As the scientific community sprints to develop diagnostic and therapeutic approaches to the virus, it is more important than ever that assays and reagents are well characterized, validated, and provide consistent, reproducible results. Cell Signaling Technology (CST) offers critical reagents (including those specifically in our SARS-CoV-2 portfolio), kits and services to support infectious disease researchers all over the world to understand how the virus behaves, how to block it, and how to cure it. CST researchers are actively engaged in scientific collaborations to understand the SARS-CoV-2 mechanism of action as well as provide insight critical in understanding the molecular machinations of the virus to be used in future vaccine development.
SARS-CoV-2 research is urgent and critical, and here at CST, we understand that the need for research antibody validation and reproducibility is paramount. There is no time to waste on reagents that produce ambiguous results, and even less on failed experiments due to poor specificity. To ensure quality, specificity, and consistency, CST adheres to the Hallmarks of Antibody Validation™ (adapted from Uhlen, M., Bandrowski, A., Carr, S. et al. A proposal for validation of antibodies. Nat Methods 13, 823–827 (2016)).
Every CST developed antibody undergoes rigorous in-house application-specific validation testing customized according to the target and needs of the individual antibody. The CST approach has always been to produce (we manufacture over 95% of our commercial products in-house) and rigorously validate our products in-house. Validation testing can include any combination of the following because no single assay alone can determine validity:
1) Binary Model - analysis of a panel of positive and negative expressing cell lines, WT vs genetic or CRISPR-based knockout
2) Ranged expression - analysis of a large panel of cell lines with known target expression levels (high vs low expressors), siRNA and heterozygous knockout
3) Orthogonal Data - verification by non-antibody-based assays (Including mass spectrometry)
4) Multiple Antibodies - Using enrichment-based applications (IP, ChIP, ChIP-seq) to compare signal observed with distinct antibodies binding to the same target or using immunostaining applications to demonstrate identical staining patterns or target localization
5) Heterologous Expression - evaluation in cell lines following heterologous expression of native (or mutated) target protein
6) Complementary Assays - confirmation of signal specificity using competitive ELISA, peptide dot blots, protein arrays, or blocking with antigen peptide to confirm elimination of modification specificity
Additionally, CST offers a large portfolio of recombinant monoclonal antibodies. These provide superior lot-to-lot consistency and enable a consistent supply, a critical factor for researchers and healthcare scientists.
As always, the results and quality of your research experiments are dependent on the inputs to draw meaningful and impactful insights. During a period of worldwide crisis, it’s crucial to use reagents you can trust to work as expected, every time, again and again. Make sure you perform your research with well validated antibodies, which can help you unlock the biological mechanisms of (SARS-CoV-2) viral disease faster and more reliably. Here at CST, we share the same goal: to help accelerate and advance research and development of assays and therapeutic drugs to take action to fight the health crisis.
References:
- https://www.bloomberg.com/amp/news/articles/2020-04-07/new-test-hopes-dashed-as-u-k-finds-antibody-kits-don-t-deliver
- https://www.genomeweb.com/molecular-diagnostics/congressional-leaders-seek-coronavirus-test-information-fda#.XpiS3lNKhTY
- https://www.cnn.com/2020/04/14/health/antibody-test-explainer/index.html
- https://www.cnn.com/2020/04/14/health/coronavirus-antibody-tests-scientists/index.html