The central nervous system (CNS) is composed of highly specialized cells, each of which play a key role in the processing of sensory and motor inputs. Neurons, one CNS cell type, specialize in the transmission and processing of electrical signals.
In addition to neurons, the CNS is also composed of non-neuronal cell types that help protect neurons and ensure proper function, such as glia. In the human brain, glia are as prevalent as neurons. Most glial cells in the human brain are oligodendrocytes (45–75%) or astrocytes (19–40%). Less than 10% are microglia or ependymal cells. However, what they lack in abundance, they make up for in significance—microglia play a critical role in neuroinflammation and neurodegenerative diseases.
View the interactive Neuronal & Glial Cell Marker Atlas for a complete list of CST antibodies.
This blog post guide includes some of the most common markers used to detect and study neuronal and glial cells.
Neuronal Markers
All neurons of the mammalian nervous system originate from neural stem cells, which are characterized by self-renewal and multipotency. Evidence suggests that aggregated proteins in the brain or exposure to long-term stimuli can result in chronic inflammation that is harmful to cells and may contribute to the pathogenesis of neurodegenerative diseases such as Alzheimer’s disease (AD), Parkinson’s disease (PD), and amyotrophic lateral sclerosis (ALS).
Detection and expression of mature neurons plays an important role in understanding the health of the CNS and can be accomplished by observing the expression of MAP2, NeuN, Neurofilament-H, Neurofilament-L, and Neurofilament-M. Neurofilament-L, for example, is a well-established CSF biomarker for neurodegeneration.1

Detection of Neurofilament-L protein in mouse brain, HeLa cells (negative control), and rat brain examined using the PathScan® Total Neurofilament Sandwich ELISA Kit #99175.
Glial Cell Markers
Microglia Markers
Microglia are immune cells that are of myeloid origin and migrate to the CNS during development. The many functions of microglia include the pruning of complex neural networks during embryogenesis, assisting in synaptic plasticity or preventing excitotoxicity by responding to neurotransmitter release, and mounting an inflammatory response to an acute infection, pathological changes, or trauma to the brain.
Specific markers for the identification of these resident macrophages of the CNS include integrin alpha M (ITGAM/CD11b), the surface glycoprotein F4/80, CD45, Iba1, and TMEM119.
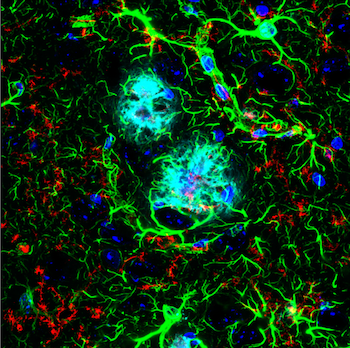 IF analysis of an amyloid mouse model brain. TMEM119 (E4B9S) Mouse mAb #98778 (red) recognizes microglia surrounding an amyloid plaque, stained with β-Amyloid (D54D2) XP® Rabbit mAb (Alexa Fluor® 647 Conjugate) #42284 (cyan pseudocolor). GFAP (E4L7M) XP® Rabbit mAb #80788 (green) detects astrocytes.
IF analysis of an amyloid mouse model brain. TMEM119 (E4B9S) Mouse mAb #98778 (red) recognizes microglia surrounding an amyloid plaque, stained with β-Amyloid (D54D2) XP® Rabbit mAb (Alexa Fluor® 647 Conjugate) #42284 (cyan pseudocolor). GFAP (E4L7M) XP® Rabbit mAb #80788 (green) detects astrocytes.
In disease states, distinct microglial populations can be identified by molecular signatures called disease-associated microglia (DAMs). DAMs mature in stages, both of which can be characterized by the expression of different biomarkers:
- Stage 1 DAM microglia upregulate TREM2 (triggering receptor expressed on myeloid cells 2), ApoE, and DAP12 and downregulate P2RY12 and transmembrane protein 119 (TMEM119).
- Stage 2 DAMs upregulate osteopontin/SPP1 and cystatin 7 (CST7).
Oligodendrocyte Markers
Oligodendrocytes are a specialized glial cell type in the CNS that produce myelin, which provides a protective sheath around the axons and improves the conduction velocity of signals between neurons. Oligodendrocytes are characterized by the expression of the myelin family proteins: myelin oligodendrocyte glycoprotein (MOG), myelin-associated glycoprotein (MAG), and Myelin Basic Protein (MBP). Another key oligodendrocyte protein, Olig2, is a useful marker for both primary and mature oligodendrocytes.
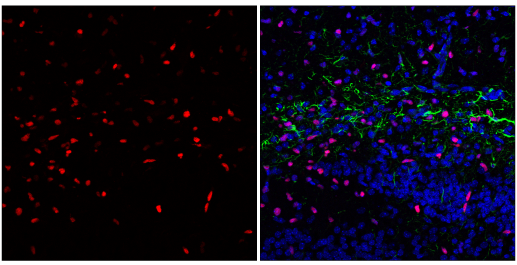 IF analysis of fixed frozen postnatal day five (P5) mouse hippocampus, labeled with Olig2 (E6G6Q) XP® Rabbit mAb #65915 (left, red) and co-labeled with GFAP (GA5) Mouse mAb (Alexa Fluor® 488 Conjugate) #3655 (right, green) and DAPI #4083 (right, blue).
IF analysis of fixed frozen postnatal day five (P5) mouse hippocampus, labeled with Olig2 (E6G6Q) XP® Rabbit mAb #65915 (left, red) and co-labeled with GFAP (GA5) Mouse mAb (Alexa Fluor® 488 Conjugate) #3655 (right, green) and DAPI #4083 (right, blue).
Oligodendrocyte progenitor cells exist in the brain to facilitate the regeneration of cells in response to injury. However, the breakdown of myelin and the inability to regenerate oligodendrocytes, along with insufficient or dysfunctional myelin have been correlated with the pathogenesis of several neurodegenerative diseases including AD, PD, ALS, and multiple sclerosis (MS).
Astrocyte Markers
Astrocytes are another immunocompetent glial cell that arise within the CNS and are found within the brain. In a healthy nervous system, astrocytes play essential roles in development, the regulation of blood flow (by supporting endothelial cells in the blood-brain barrier), synaptic transmission and function, and energy and metabolism (by providing nutrients to neurons and synthesizing certain neurotransmitters). The most specific marker for astrocytes is GFAP. 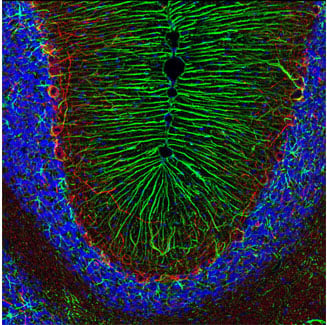
IF analysis of rat cerebellum using GFAP (D1F4Q) XP® Rabbit mAb #12389 (green) and Neurofilament-H (RMdO 20) Mouse mAb #2836 (red). Blue pseudocolor = DRAQ5® #4084 (fluorescent DNA dye).
S100B is abundantly expressed in astrocytes as well. However, it is also selectively found in myelinating oligodendrocytes. Elucidating the expression and function of S100B is an active area of research.
Loss of function or abnormal function of astrocytes has been implicated in a wide variety of neurodegenerative disease processes. Chronic activation of astrocytes results in the formation of lesions similar to those observed in AD and Huntington's disease.
Antibodies to Advance Neuroscience Research
Highly specific antibodies that recognize and distinguish neuronal and glial cell types are critical for advancing our understanding of neuroscience. Cell Signaling Technology enables you to characterize mixed cell populations with confidence.
Find the right cell-type markers for your next experiment using the interactive Neuronal and Glial Cell Marker Atlas.
Additional Resources
- Neurodegenerative disease pathways:
Select References
-
Gaetani L, Blennow K, Calabresi P, Di Filippo M, Parnetti L, Zetterberg H. Neurofilament light chain as a biomarker in neurological disorders. J Neurol Neurosurg Psychiatry. 2019;90(8):870-881. doi:10.1136/jnnp-2018-320106






/Binary_oasis_Blog%20Header.webp)
