Tau is a microtubule-associated protein expressed predominantly in the parts of the brain that function to stabilize axonal microtubules. Tau exists as eight different isoforms and can be phosphorylated at a number of unique sites, which, in turn, decreases its ability to bind microtubules. While Tau is phosphorylated in normal, healthy brain, it becomes hyperphosphorylated in neurodegenerative diseases, including Alzheimer’s disease (AD).
Role of Tau Protein and Neurofibrillary Tangles in Alzheimer’s Disease
Neurofibrillary tangles found in Alzheimer’s disease result from the hyperphosphorylation of tau. Phosphorylation of tau can occur at many sites, typically by the kinases GSK3 and CDK5, and results in a decreased ability to bind microtubules.
 |
Explore the interactive Molecular & Cellular Biology of Alzheimer’s Disease Pathway Diagram to see key protein targets and associated CST products. |
Hyperphosphorylation results in tau dissociation and subsequent destabilization of the microtubules and oligomerization of tau. Neurofibrillary tangles, as observed in Alzheimer’s disease and other tauopathies, are collections of paired helical filaments composed of hyperphosphorylated tau. It is thought that tau proteins are able to form prion-like “seeds” by which it can spread from neuron to neuron, and the presence of neurofibrillary tangles eventually leads to neuronal apoptosis and further pathogenesis of disease.
 |
Explore the Regulation of Apoptosis Signaling Pathway to explore key protein targets and associated CST products. |
Monitoring Phosphorylated and Total Tau
The levels of total and phosphorylated tau can be used to assist in the diagnosis and treatment of neurodegeneration. For example, total and phosphorylated tau are highly sensitive biomarkers that can predict the progression of mild cognitive impairment to Alzheimer’s disease.
The phosphorylation status of tau dramatically affects the intracellular distribution of the protein. Phosphorylated tau has been found in the neuronal soma of hippocampal neurons as well as immortalized GnRH neurons. Further evidence demonstrates that tau phosphorylation at Ser404 destabilizes microtubules as well as a link between Alzheimer’s disease and hyperphosphorylation at Ser404.
Importance of Antibodies in Tau Detection
Monoclonal antibodies to tau are designed to recognize endogenous levels of tau protein as well as various forms of phosphorylated tau including: Thr217, Thr181, Ser199, Ser202, Thr205, Ser214, Thr231, Ser396, Ser404, and Ser416. The levels of tau aggregated in neurofibrillary tangles can be determined in a tissue sample through western blotting, immunoprecipitation, immunofluorescence, ELISA, immunohistochemistry.
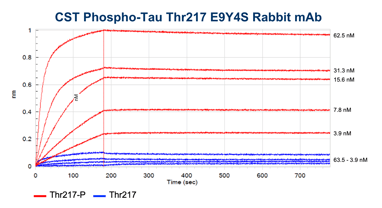
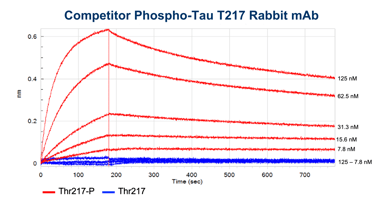
BLI data comparing the CST antibody clone (left) to a competitor monoclonal antibody (right). Binding to the p-tau217 clone E9Y4S peptide is shown in red and binding to the non-phosphorylated peptide in blue. Concentrations tested range from 250 nM to 3.9 nM. Global curve-fitting was performed using a bivalent analyte model; only the concentrations that passed that fitting are shown. From the blog: Unraveling Alzheimer’s Disease: Developing a P-Tau217 Antibody
In support of neurodegeneration research, a number of antibodies have been developed by scientists at Cell Signaling Technology that enable the analysis of tau and phosphorylated tau.







