Getting started with flow cytometry or beginning to branch out from working solely with live cells to fixation and permeabilization protocols can be a daunting yet rewarding undertaking. You can gain insight into differences in not only surface protein expression, but also intracellular protein expression, phosphorylation status, and more. Combining these types of readouts can provide unique understandings of cellular populations and their signaling profiles all at once.
Given the wide range of protocols available, we developed an Antibody Staining Guide for Flow Cytometry, available globally for researchers to download, to help demystify the antibody staining process and guide you through your assay set-up step-by-step. Using the decision tree steps within the Antibody Staining Guide and this blog post, we aim to address some of the common questions you may have.
How do I stain surface proteins for flow cytometry experiments?
Let's start out by walking through a scenario in which you are interested in staining mouse splenocytes with a pair of surface proteins, CD4 and TCRβ. There are mouse reactive flow-validated antibody conjugates available against each target: CD4 and TCRβ. (Note: Clone H57-597 clone, which targets TCRβ, and is discussed in this blog, has been discontinued.) While you know that each protein is found at the plasma membrane, you need to check whether the antibodies recognize extracellular or intracellular portions of each.
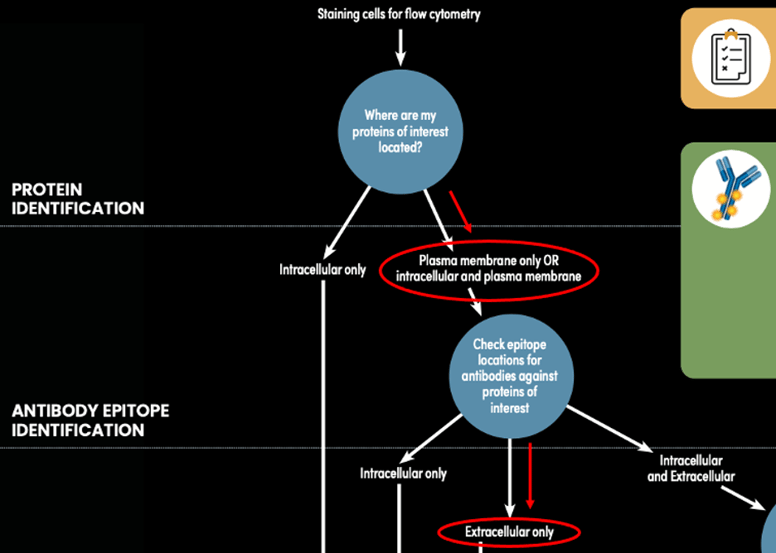 Figure 1: Determining the appropriate staining protocol path for the CD4 (RM4-5) and TCRβ (H57-597) example, using the Antibody Staining Guide for Flow Cytometry (represented by red lines and circles).
Figure 1: Determining the appropriate staining protocol path for the CD4 (RM4-5) and TCRβ (H57-597) example, using the Antibody Staining Guide for Flow Cytometry (represented by red lines and circles).
The product webpage for each CST antibody includes a Source / Purification section where basic information about the antigen can be found. This may list the N- or C-terminal part of the protein or a central amino acid, or the page may simply say that the antibody detects an epitope within an extracellular or intracellular domain. If you are ever unsure, contact technical support for more information.
Referencing the product pages, the RM4-5 clone, targeting CD4, and the H57-597 clone, targeting TCRβ (product discontinued), both detect the extracellular domains of their respective targets. This means you can stain live cells using both antibodies at the same time. Since you are multiplexing two antibodies in the same experiment, you need to choose fluorophores with different excitation/emission profiles to avoid or minimize the need for compensation. You decide to go with APC and FITC. Here's how your results look putting it all together and staining live mouse splenocytes with CD4 (RM4-5) Rat mAb (APC Conjugate) and TCRβ (H57-597) Hamster mAb (FITC Conjugate) (product discontinued):
 Figure 2: Analysis of C57BL/6 spleen cells stained with TCRβ (H57-597) Hamster mAb (FITC Conjugate) (right) or concentration-matched Armenian Hamster Isotype Control (FITC Conjugate) (left) using a live cell protocol. Samples were co-stained with CD4 (RM4-5) Rat mAb (APC Conjugate).
Figure 2: Analysis of C57BL/6 spleen cells stained with TCRβ (H57-597) Hamster mAb (FITC Conjugate) (right) or concentration-matched Armenian Hamster Isotype Control (FITC Conjugate) (left) using a live cell protocol. Samples were co-stained with CD4 (RM4-5) Rat mAb (APC Conjugate).
How do I stain surface and intracellular proteins in the same sample for flow cytometry experiments?
Of course, gathering data about cell surface protein expression is great, but you may also want information about what's going on inside the cell. Generally, knowing surface protein expression profiles allow you to identify the types of cells present in your sample, while examining intracellular proteins and post-translational modifications can provide insight into how the cells are functioning. In this scenario, you are interested in Tox/Tox2 expression in T cells (which you can identify in a peripheral blood mononuclear cell (PBMC) population by CD3 expression). Since you are probing for an intracellular protein (Tox/Tox2), you know that fixation and permeabilization will be required.
Rather than resigning yourself to devoting extra time in the lab to stain your sample sequentially, you first check whether a CD3 co-stain can be used on fixed and permeabilized cells by consulting the CST Protocol Compatibility Table.
The human-reactive CD3 clone, UCHT1, which recognizes an extracellular epitope, works well in all of the fixation and permeabilization protocols commonly recommended by CST. This means you will be able to save some time by staining PBMCs simultaneously with the anti-CD3 (UCHT1) and anti-Tox/Tox2 (E6G5O) antibodies, after fixation and permeabilization. To fix and permeabilize, you can use the 4% formaldehyde fixation and 90% ice cold methanol permeabilization protocol recommended for the Tox/Tox2 antibody. The reagents are all available individually or in the Intracellular Flow Cytometry Kit (Methanol) #13593.
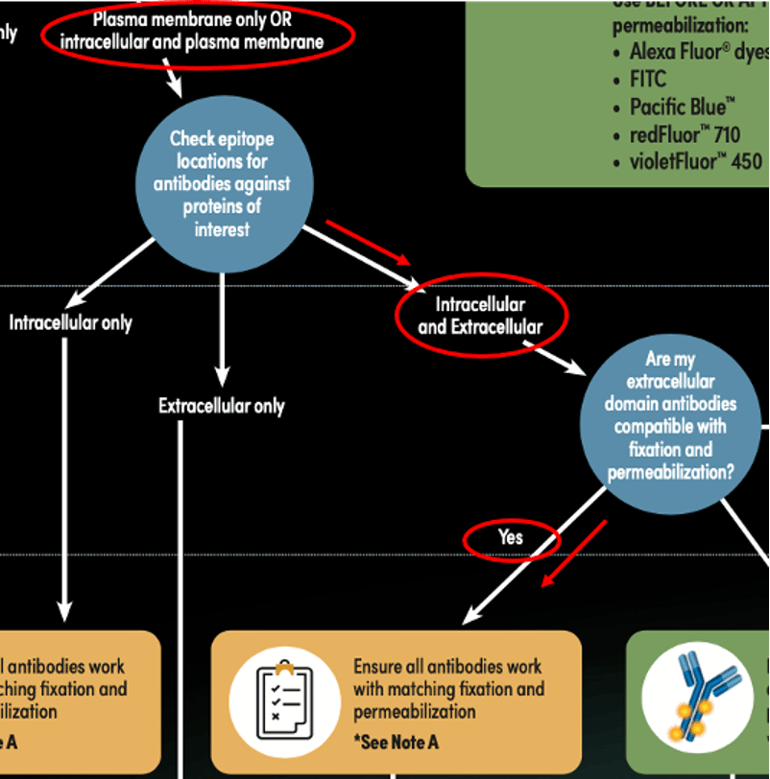 Figure 3: Determining an appropriate staining protocol path for the CD3 (UCHT1) and Tox/Tox2 (E6G5O) example, using the Antibody Staining Guide for Flow Cytometry (represented by red lines and circles).
Figure 3: Determining an appropriate staining protocol path for the CD3 (UCHT1) and Tox/Tox2 (E6G5O) example, using the Antibody Staining Guide for Flow Cytometry (represented by red lines and circles).
Here's how the sample looks -- you have distinct populations of PBMCs that fall into each of the four quadrants:
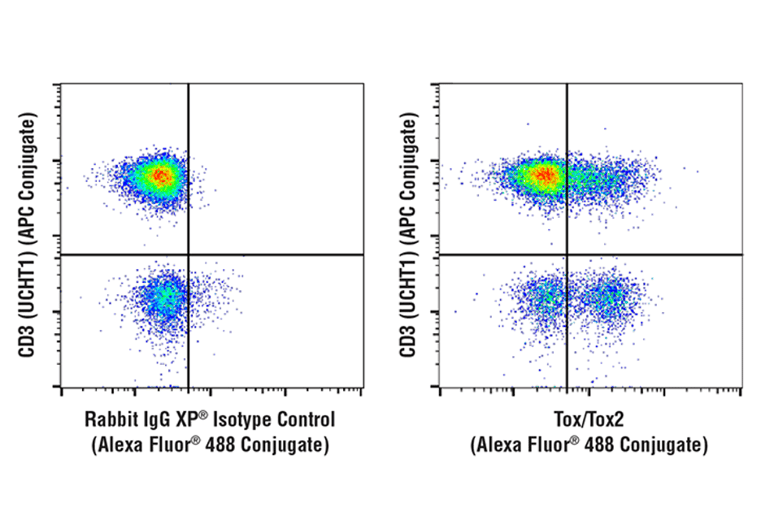 Figure 4: Analysis of fixed and permeabilized human PBMCs, gated on the lymphocyte population, stained with Tox/Tox2 (E6G5O) Rabbit mAb (Alexa Fluor® 488 Conjugate) (right) or concentration-matched Rabbit (DA1E) mAb IgG XP® Isotype Control (Alexa Fluor® 488 Conjugate). Samples were co-stained with CD3 (UCHT1) Mouse mAb (APC Conjugate).
Figure 4: Analysis of fixed and permeabilized human PBMCs, gated on the lymphocyte population, stained with Tox/Tox2 (E6G5O) Rabbit mAb (Alexa Fluor® 488 Conjugate) (right) or concentration-matched Rabbit (DA1E) mAb IgG XP® Isotype Control (Alexa Fluor® 488 Conjugate). Samples were co-stained with CD3 (UCHT1) Mouse mAb (APC Conjugate).
What to do When Surface and Intracellular Proteins Can't be Stained Simultaneously
Unfortunately, there are cases where you will not be able to stain samples with all your antibodies simultaneously. In this next example, you want to determine the fraction of CD11c-positive PBMCs that express the transcription factor BATF3. Like before, you are interested in an intracellular target and a membrane target.
This time, the Protocol Compatibility Table shows you that the human reactive CD11c (3.9) clone performs adequately on live cells but does not perform well on cells that have been fixed and permeabilized with any protocol other than the FoxP3/Transcription Factor Protocol. You also have another consideration: there are no direct conjugates available OTS for the BATF3 (E3K5H) clone you have selected. (Note: OTS conjugates for the BATF3 (E3K5H) clone were made available after this blog was published. View here: BATF3 (E3K5H) clone). Fortunately, you are only using one co-stain (CD11c), and it was raised in a different animal (mouse) than the BATF3 antibody (rabbit), so you will be able to use an anti-rabbit secondary antibody to detect BATF3.
Since the BATF3 antibody product page recommends using formaldehyde fixation followed by methanol permeabilization, you plan to first stain live PBMCs with the CD11c antibody and then follow the protocol recommended for BATF3.
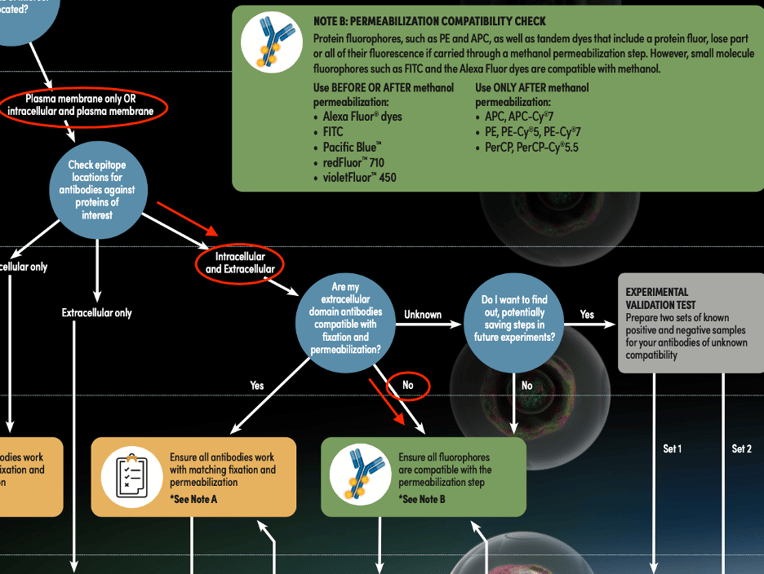 Figure 5: Determining the appropriate staining protocol path for the CD11c (3.9) and BATF3 (E3K5H) example, using the Antibody Staining Guide for Flow Cytometry (represented by red lines and circles).
Figure 5: Determining the appropriate staining protocol path for the CD11c (3.9) and BATF3 (E3K5H) example, using the Antibody Staining Guide for Flow Cytometry (represented by red lines and circles).
Checking the callout in the decision tree, you see that methanol is an organic solvent that can denature protein fluorophores, so you make sure to choose a CD11c antibody conjugated to a small molecular fluorophore, in this case, FITC, which will retain its fluorescence when carried through a methanol permeabilization step. After the sample has been labeled with the CD11c FITC conjugate, you fix and permeabilize the sample, stain the sample with the BATF3 antibody, and then stain with an anti-rabbit Alexa Fluor® 647 conjugated secondary antibody. Once you have run your sample and analyzed the data, the result is a clear separation of positive and negative populations for both readouts:
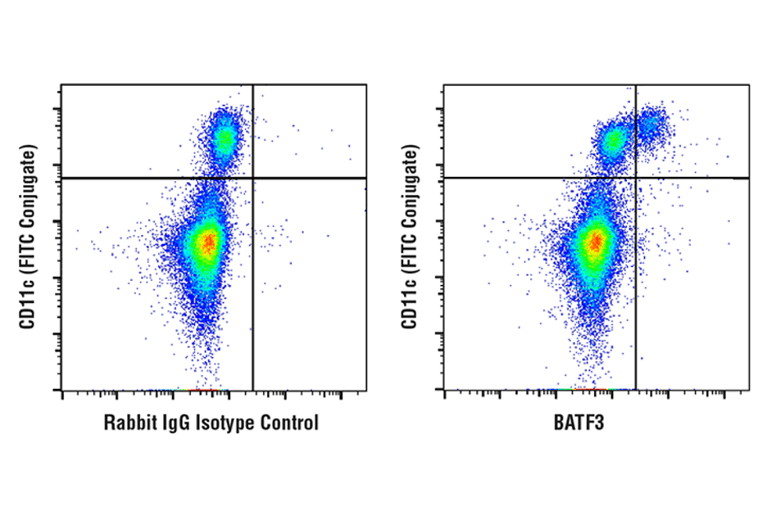 Figure 6: Analysis of fixed and permeabilized human PBMCs using BATF3 (E3K5H) Rabbit mAb (right) or concentration-matched Rabbit (DA1E) mAb IgG XP® Isotype Control (left), co-stained prior to fixation with CD11c (3.9) Mouse mAb (FITC Conjugate). Anti-rabbit IgG (H+L), F(ab')2 Fragment (Alexa Fluor® 647 Conjugate) was used as a secondary antibody.
Figure 6: Analysis of fixed and permeabilized human PBMCs using BATF3 (E3K5H) Rabbit mAb (right) or concentration-matched Rabbit (DA1E) mAb IgG XP® Isotype Control (left), co-stained prior to fixation with CD11c (3.9) Mouse mAb (FITC Conjugate). Anti-rabbit IgG (H+L), F(ab')2 Fragment (Alexa Fluor® 647 Conjugate) was used as a secondary antibody.
In the scenarios described here, we referenced the Antibody Staining Guide for Flow Cytometry and the Protocol Compatibility Table to guide situations where samples were stained with two antibodies, but you can also use these resources to guide your decisions when multiplexing with three or more readouts. Regardless of whether you are staining live cells, fixed and permeabilized cells, or a combination of both, be sure to take general flow cytometry considerations into account, such as including proper controls and minimizing spectral overlap of your fluorophores.
Finally, if you need additional flexibility with your fluorophores, consider inquiring about CST custom antibody conjugation services. Good luck with your experiments!






/Binary_oasis_Blog%20Header.webp)
