Pancreatic cancer is devastating and difficult to diagnose early. The organ is located deep inside the body, so tumors aren’t detected during routine physical exams, and patients are usually asymptomatic until the cancer is large and, oftentimes, metastatic.1 The five-year survival rate for localized pancreatic cancer is 42%, however, this number plummets to 3% once the cancer has metastasized.2 Unfortunately, up to 80% of all pancreatic cancer cases are diagnosed in later stages, and many patients succumb to the disease within a year.
Pancreatic Cancer Biomarkers and Drivers
There is an urgent need for new methods to diagnose and treat pancreatic cancer, particularly pancreatic ductal adenocarcinoma (PDAC), which constitutes the vast majority of pancreatic cancer cases and is the most difficult to treat. Biomarkers like CA 19-9 and MUC5AC are being explored as prognosis indicators and have potential as diagnostic biomarkers when used in combination.3
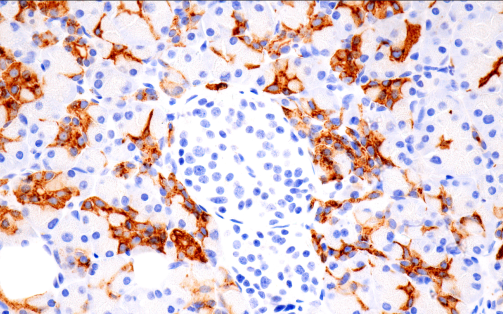
Immunohistochemical analysis of paraffin-embedded normal human pancreas using CA 19-9 (C241:5:1:4) Mouse mAb #68030 performed on the BOND RX Leica Biosystems.
PDAC also displays high levels of desmoplasia, or the formation of connective tissue in response to tumors, so fibrotic markers like α-smooth muscle actin, COL1A1, and Fibronectin/FN1 may also potentially be used in research to track treatment response. Loss-of-function mutations in the tumor suppressors p16 INK4A, p53, and SMAD4 and gain-of-function of mutations in K-Ras are frequently observed in PDAC.
Studying these mutations can deliver further insights into the molecular mechanisms driving disease progression and can potentially help identify new therapeutic targets and approaches.
Targeting the Tumor Microenvironment
A remarkable feature of PDAC is the abundance of extracellular matrix (ECM) found within the tumor mass, in some cases constituting as much as 90% of the tumor volume. The ECM molecules constitute an important part of the tumor microenvironment (TME) and can significantly influence tumor development. Thus, a promising, novel therapeutic approach for pancreatic cancer is to manipulate the TME using therapies targeting components of the TME, such as metabolic substrates, ECM macromolecules, and/or components of the immune system. For example, studies have shown that targeting macrophages and granulocytes with CSF-1R or CXCR2 inhibitors sensitizes PDAC tumors to immune checkpoint inhibitors.4
One way to manipulate the TME is to target the function and behavior of cancer-associated fibroblasts (CAFs), the cell type largely responsible for the fibrotic TME. CAFs are a heterogeneous cell population; there are subsets with tumor-promoting functions, in addition to subsets that can suppress tumor development. Tumor-restrictive myofibroblastic CAFs (myCAFs) are contractile and express alpha-smooth muscle actin. Tumor-promoting inflammatory CAFs (iCAFs) secrete cytokines like IL-6 and LIF and promote survival, motility, and cell migration, all processes that can contribute to tumor metastasis.
TGFβ signaling in CAF cells near tumor cells promote myCAF features, while IL1a secretion by the tumor through the Jak/Stat pathway promote iCAF features. Studies show that Oncostatin M (OSM) signaling through the Oncostatin M Receptor (OSMR) can reprogram CAFs to a pro-tumorigenic nature. Additionally, tumor cells implanted in OSM-deficient mice grew slower and did not metastasize, delaying disease progression.
Finally, targeting CAFs with Vitamin D Receptor agonists, LIF-blocking antibodies or CXCR4 inhibitors has been shown to improve the response to some therapies, including both chemotherapies and immunotherapies. This suggests that, because of the fibrotic nature of PDAC tumors, targeting the TME is another therapeutic approach to potentially improve the clinical outcome for pancreatic cancer patients.
Additional Resources
Explore the CST Cancer Research resource center for access to related webinars, pathways, and guides.
Select References
- American Cancer Society – Key Statistics for Pancreatic Cancer https://www.cancer.org/cancer/pancreatic-cancer/about/key-statistics.html
- John Hopkins Medicine – Pancreatic Cancer Prognosis https://www.hopkinsmedicine.org/health/conditions-and-diseases/pancreatic-cancer/pancreatic-cancer-prognosis
- Zhang, J., Wang, Y., Zhao, T. et al.Evaluation of serum MUC5AC in combination with CA19-9 for the diagnosis of pancreatic cancer. World J Surg Onc 18, 31 (2020). https://doi.org/10.1186/s12957-020-1809-z.
- Lee BY, Hogg EKJ, Below CR, et al. Heterocellular OSM-OSMR signalling reprograms fibroblasts to promote pancreatic cancer growth and metastasis. Nat Commun. 2021;12(1):7336. Published 2021 Dec 17. doi:10.1038/s41467-021-27607-8





/Binary_oasis_Blog%20Header.webp)
